Prognostic capabilities like being able to detect the development of dementia, would give patients and their families time to plan and manage treatment and care. It would also give care takers the ability to focus time and energy on those people most likely to develop dementia. Thanks to artificial intelligence research conducted at McGill University, this kind of predictive power could soon be available to clinicians everywhere.
Dr. Pedro Rosa-Neto, co-lead author of the study and Associate Professor in McGill’s departments of Neurology & Neurosurgery and Psychiatry, expects that the algorithm will change the way physicians manage patients and greatly accelerate treatment research into Alzheimer’s disease. “This is an example how big data and open science brings tangible benefits to patient care,” says Dr. Rosa-Neto (also director of the McGill University Research Centre for Studies in Aging).
Clinical trials could focus only on individuals with a higher likelihood of progressing to dementia within the time frame of the study.” This will greatly reduce the cost and the time necessary to conduct these studies,” adds Dr. Serge Gauthier, co-lead author and Professor of Neurology & Neurosurgery and Psychiatry at McGill.
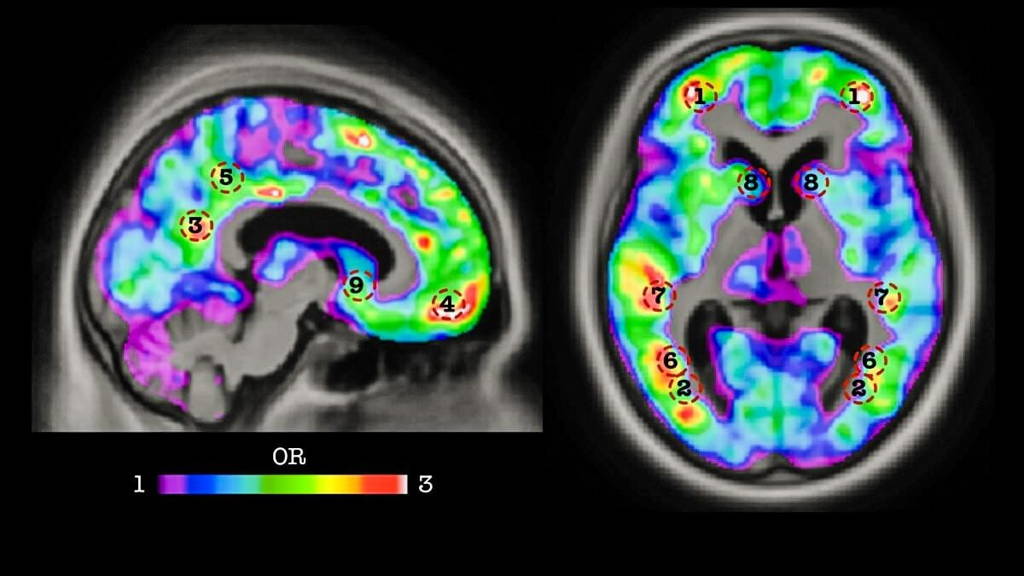
Hundreds of amyloid PET scans of MCI patients from the ADNI database were used to train the team’s algorithm to identify which patients would develop dementia, with an accuracy of 84 percent, before symptom onset. Research is ongoing to find other biomarkers for dementia that could be incorporated into the algorithm in order to improve the software’s prediction capabilities.
While new software has been made available online to scientists and students, physicians won’t be able to use this tool in clinical practice before certification by health authorities. To that end, the McGill team is currently conducting further testing to validate the algorithm in different patient cohorts, particularly those with concurrent conditions such as small strokes.
Also in 2016, T-Mobile and Alzheimer Netherlands introduced the Dutch version of the game ‘Sea Hero Quest’ for iOS and Android. The game lets users find their way through a maze and has been developed to support research into Alzheimer's disease. The game makes it possible for researchers to gather information such as data on how the brain works whilst navigating through a maze. Two minutes of game time represents five hours of lab research. The games had before been introduced in the US, the UK and Germany, where it has been downloaded over a million times. According to Marc Petit, director scientific research with Alzheimer Netherlands, the game offers a good 'research at home' alternative for the often time consuming and expensive lab research into the way the human brain works.
Early recognition of dementia signatures
Scientists from McGills Douglas Mental Health University Institute’s Translational Neuroimaging Laboratory, used AI- and big data techniques to develop an algorithm capable of recognizing the signatures of dementia two years before its onset, using a single amyloid PET scan of the brain of patients at risk of developing Alzheimer’s disease. Their findings appear in a new study published in the journal Neurobiology of Aging.Dr. Pedro Rosa-Neto, co-lead author of the study and Associate Professor in McGill’s departments of Neurology & Neurosurgery and Psychiatry, expects that the algorithm will change the way physicians manage patients and greatly accelerate treatment research into Alzheimer’s disease. “This is an example how big data and open science brings tangible benefits to patient care,” says Dr. Rosa-Neto (also director of the McGill University Research Centre for Studies in Aging).
Clinical trials could focus only on individuals with a higher likelihood of progressing to dementia within the time frame of the study.” This will greatly reduce the cost and the time necessary to conduct these studies,” adds Dr. Serge Gauthier, co-lead author and Professor of Neurology & Neurosurgery and Psychiatry at McGill.
Amyloid as biomarker of dementia
Scientists have long known that a protein known as amyloid accumulates in the brain of patients with mild cognitive impairment (MCI), a condition that often leads to dementia. Though the accumulation of amyloid begins decades before the symptoms of dementia occur, this protein couldn’t be used reliably as a predictive biomarker because not all MCI patients develop Alzheimer’s disease. To conduct their study, the McGill researchers drew on data available through the Alzheimer’s Disease Neuroimaging Initiative (ADNI), a global research effort in which participating patients agree to complete a variety of imaging and clinical assessments.Hundreds of amyloid PET scans of MCI patients from the ADNI database were used to train the team’s algorithm to identify which patients would develop dementia, with an accuracy of 84 percent, before symptom onset. Research is ongoing to find other biomarkers for dementia that could be incorporated into the algorithm in order to improve the software’s prediction capabilities.
While new software has been made available online to scientists and students, physicians won’t be able to use this tool in clinical practice before certification by health authorities. To that end, the McGill team is currently conducting further testing to validate the algorithm in different patient cohorts, particularly those with concurrent conditions such as small strokes.
Growing number of studies into causes, prevention
There has been a growing number of studies into the causes of dementia and earlier detection. Last year, a team of neuroscience and biochemistry researchers at Stony Brook University made a discovery that illustrates for the first time the difference between amyloid buildup in brain blood vessels and amyloid buildup around brain neurons. Their findings may provide a new path to research on Alzheimer’s disease and its cause.Also in 2016, T-Mobile and Alzheimer Netherlands introduced the Dutch version of the game ‘Sea Hero Quest’ for iOS and Android. The game lets users find their way through a maze and has been developed to support research into Alzheimer's disease. The game makes it possible for researchers to gather information such as data on how the brain works whilst navigating through a maze. Two minutes of game time represents five hours of lab research. The games had before been introduced in the US, the UK and Germany, where it has been downloaded over a million times. According to Marc Petit, director scientific research with Alzheimer Netherlands, the game offers a good 'research at home' alternative for the often time consuming and expensive lab research into the way the human brain works.